It is no secret that cell-based models are the backbone of modern drug discovery. For decades, researchers have used animal-based models including cells and whole animals to investigate mechanisms of disease, screen compounds, and support preclinical research. Over the past two decades, advances in human primary cell culture, induced pluripotent stem cells (iPSCs), gene editing, and 3D culture have expanded the range of human-relevant in vitro models available. Many of these emerging technologies are now (as of 2026) discussed within the context of new approach methodologies (NAMs), which includes methods that aim to advance research and development while reducing the use of animals (Reviewed in 1).
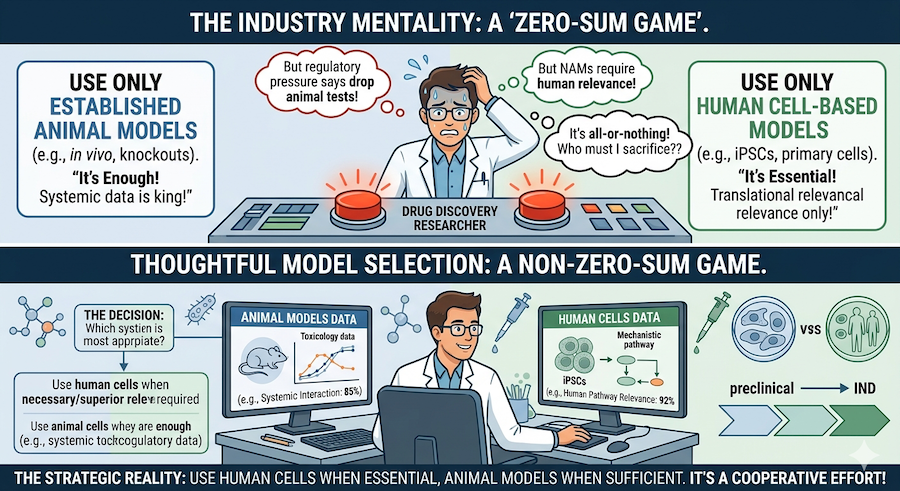
As interest in NAMs grows, researchers may feel they should drop animal models completely in favor of human cell-based alternatives and/or AI/ML algorithm-driven approaches. This reflects the scientific advances that make it possible to use non-animal models, as well as growing regulatory and societal pressure to reduce dependence on animal testing. For example, the FDA and EMA now encourage the development and use of NAMs, and recent policy changes make it possible to use non-animal methods in place of traditional animal studies in some contexts (2). However, rather than an ‘all or nothing’ 0:1 outcome, we believe the key question is which model is most appropriate for the stage of research and the scientific question being asked and how we can reduce unnecessary/excessive use of animal models.
In this article, we summarize the strengths and limitations of animal- and human-derived cells, how new technologies have created inflection points in drug discovery, and why moving toward more human-relevant models is best approached in a stepwise strategic manner rather than an overnight replacement.
Strengths and limitations of animal-based models
For decades, the widespread use of animal models has reflected a long history of refining these systems through genetic engineering, assay optimization, and experimental standardization. One major advantage of animal models and animal-derived cell models is the vast body of scientific literature that supports them, which gives researchers access to a wide range of well-characterized models, including knockout, knock-in, and transgenic animal cells and whole animals designed to investigate specific genes, pathways, and disease phenotypes.
Animal-derived cells also offer practical advantages. They are relatively robust in culture and familiar to laboratories with established experimental workflows. Protocols are typically well optimized, and many assays are supported by extensive historical datasets describing expected phenotypes and experimental behavior. These factors often make animal cells the go-to choice for mechanistic studies, early-stage compound screening, and experiments that require reproducibility within a well-established model system.
Despite their strengths, animal-derived cells have many drawbacks, particularly when the goal is to understand or predict human biology. The most obvious issue is species difference. Cells from mouse, rat, dog, or non-human primate origin do not always respond to compounds, stressors, or disease-associated perturbations in the same way as human cells. For example, differences in receptor biology, signaling pathways, ion channels, transporter expression, metabolism, and immune responses (or, how they react to laboratory cell culture flasks!) can all influence experimental outcomes.
This is critical because the resulting translational gap is particularly stark in neurodegeneration. While researchers have successfully “cured” human-like Alzheimer’s, amyotrophic lateral sclerosis (ALS), and multiple sclerosis (MS) in animal models, the same experimental drugs have consistently failed to provide therapeutic benefit when administered to humans in clinical trials. Animal-derived cells may therefore be ideal for some questions, but less suitable for others, particularly when human-specific biology is central to the mechanism being studied.
Human cell-based models: advantages, challenges, and new solutions
Human cell-based models offer the obvious advantage: they allow researchers to study biological processes directly in human cells. This is important in situations where species differences are likely to impact experimental outcomes, such as in neurobiology, liver metabolism, and certain aspects of immunology and toxicity. Primary human cells have long been valued for this reason. When available, they can recapitulate key features of native human cell biology and are often considered a useful reference point for in vitro studies. However, working with primary human cells is not without challenges. Some cell types are difficult to obtain in sufficient numbers, while others are difficult to maintain in culture once isolated. Human brain microvascular endothelial cells and sensory neurons are examples of cell types that are both highly relevant and notoriously difficult to source or culture reliably. Human primary Kupffer cells (liver) and LSECs (liver) are impossible to procure/extract from liver biopsies and they do NOT survive in cell culture most of the time.
Donor-to-donor variability presents another challenge. Human primary cells can behave differently depending on genetic background, age, medical history, environmental exposures, tissue quality, and isolation conditions. In some settings this variability reflects meaningful biology, but in others it introduces reproducibility challenges. There are also practical issues related to supply and consistency. In toxicology, for example, teams sometimes strongly prefer a particular lot of primary hepatocytes because it performs reliably in a given assay. Once that batch is exhausted, replacing it may introduce variability that affects data continuity. This tendency to “hoard” well-performing LOT#s highlights how fragile some primary-cell workflows can be.
One approach that has helped address some of these limitations is the use of induced pluripotent stem cell (iPSC)-derived models. Because iPSCs can be expanded and differentiated into multiple cell types, they provide a renewable source of human cells that may otherwise be rare, inconsistent, or difficult to obtain.
For drug discovery teams, the use of iPSCs can improve scalability, support assay standardization, and reduce reliance on unpredictable donor tissue supply. Are iPSC-derived cells perfect substitutes for primary human cells? No, and issues such as cellular maturity, functional equivalence, patients vs. donors (background challenges) and batch effects remain important considerations. Nevertheless, in many contexts they provide a practical middle ground: more human-relevant than animal-derived cells, and more scalable and reproducible than scarce primary human material, making iPSC technologies increasingly important within the NAMs landscape.

A stepwise strategic shift
Scientific progress is often driven by the tools available to researchers. New technologies can create inflection points that reshape what kinds of experiments become feasible. The rise of knockout and transgenic animals is a notable example here. These technologies greatly expanded the ability to study gene function and helped establish animal models as the dominant experimental system across many areas of biology.
More recent advances, including stem cell reprogramming and differentiation protocols, CRISPR-based genome editing, single-cell analysis, and organoid culture, are now expanding what can be done with human cells. Researchers increasingly have access to human-derived models that were previously unavailable, impractical, or too inconsistent to use at scale. Importantly, this doesn’t mean that older model systems become obsolete. Instead, technological advances expand the experimental toolbox and create new opportunities to study biology in different ways.
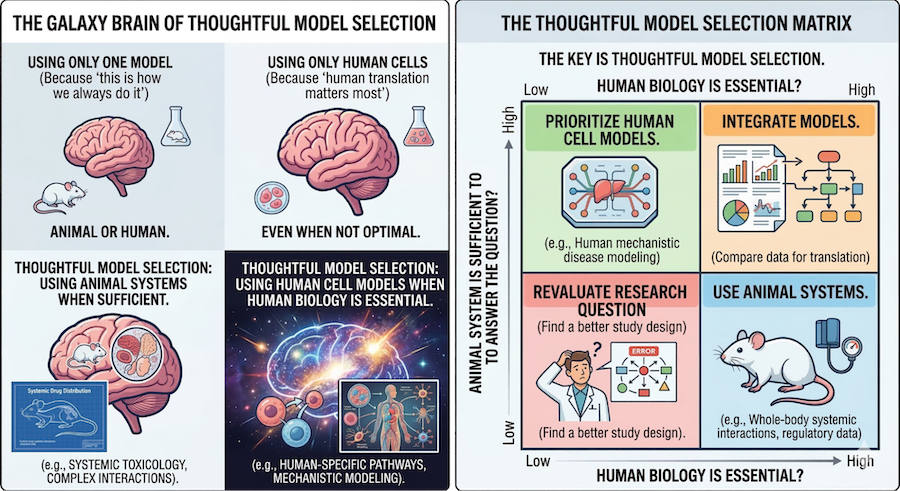
Choosing the right model: when human cells are necessary and animal systems are sufficient
Translational relevance and predictive value depend on the question being asked and the type of experiment being performed. For some disease-modeling assays, the goal may simply be to capture one or two phenotypes relevant to a mechanism of interest. In that situation, a relatively simple cell-based system may be sufficient. In other settings, such as IND-enabling studies, the expectations are much higher. For this reason, the question is not whether human cells are always better than animal cells, or vice versa. The more useful question is which model is most appropriate for the biological question being asked.
In some situations, human cells are clearly necessary, for example, when species differences are likely to impact interpretation or when the biology being studied is inherently human, e.g., a human-specific disease such as HIV. In other situations, animal-based systems may be sufficient or required, particularly if the underlying biology is conserved or the goal is a mechanistic study within a well-established model system. The key is thoughtful and stepwise model selection: use human cells when human biology is essential, and animal systems if/when they are absolutely required by regulatory authorities.
The transition toward more human-relevant systems won’t happen overnight, but the experimental toolbox has never been more diverse. Established animal-based workflows cannot simply be replaced all at once, particularly in regulated areas of drug development. Instead, progress will likely occur stepwise, through benchmarking studies, comparison experiments, and the gradual integration of new models into existing research pipelines. In our view, NAMs are not simply about replacing one system with another, but about selecting the right tool for the right stage of research. We look forward to seeing how these new technologies continue to reshape the drug discovery landscape.
Stay tuned for the next installment of this new series about NAMs!
References
- Sewell F, Alexander-White C, Brescia S, Currie RA, Roberts R, Roper C, Vickers C, Westmoreland C, Kimber I. New approach methodologies (NAMs): identifying and overcoming hurdles to accelerated adoption. Toxicol Res (Camb). 2024 Mar 25;13(2):tfae044. doi: 10.1093/toxres/tfae044.
- Han JJ. FDA Modernization Act 2.0 allows for alternatives to animal testing. Artif Organs. 2023 Mar;47(3):449-450. doi: 10.1111/aor.14503.
—
Karen O’Hanlon Cohrt is an independent Science Writer with a PhD in biotechnology from Maynooth University, Ireland (2011). After her PhD, Karen relocated to Denmark where she held postdoctoral positions in mycology and later in human cell cycle regulation, before moving to the world of drug discovery. Karen has been a full-time science writer since 2017, and has since then held numerous contract roles in science communication and editing spanning diverse topics including diagnostics, molecular biology, and gene therapy. Her broad research background provides the technical know-how to support scientists in diverse areas, and this in combination with her passion for learning helps her to keep abreast of exciting research developments as they unfold. Karen is currently based in Ireland, and you can follow her on Linkedin here.
