A friend of my family was diagnosed with Parkinson’s Disease (PD) a few years ago. She was, and still is, a very active member of her community. She hosts prayer evenings, children’s groups to teach virtues, and even volunteers at her local retirement home. One day, we were sitting down together in her small kitchen having a cup of hot earl grey tea and a good chat when I noticed she was favoring her left hand. When she sat down, she gripped her right hand in her left and as she gesticulated to animate her story, she held her right arm tightly at her waist. I mentioned what I’d seen to my mom and she told me about the diagnosis.
Some illnesses and conditions are somewhat hidden allowing us to keep up appearances. With a characteristic early symptom being a tremor of the hand, PD is not so obliging.
Like Alzheimer’s Disease and Multiple Sclerosis, PD is a chronic, progressive disorder with an unknown etiology and no cure. Therapies, surgery and medication are used to treat symptoms and sometimes delay disease progression and research into several new treatment options is ongoing. There is evidence of a genetic component and further research in this area may lead to a better understanding of what triggers this disorder.
Here, we’ll outline what we do know about PD by focusing on the presumed protagonists of the story: dopaminergic neurons.
Dopaminergic Neurons
Dopaminergic neurons are nerve cells in the substantia nigra of the midbrain, the part of the brain just above the spinal cord and below the cerebrum. These cells are our body’s main source of dopamine, ring any bells? Dopamine is a neurotransmitter involved in regulating movement and emotional responses. You might remember hearing how our brain loves to push us to behave on reward and pleasure? Well you can thank dopamine for reaction. Dopamine also plays a role in attention, mood, sleep, learning and memory. Now with all of this in mind, take a moment to think about a brain with a progressively decreasing amount of dopamine.
What do you picture?
Parkinson’s Disease
In PD, neurons in the brain begin to malfunction and die. The symptoms of PD include:
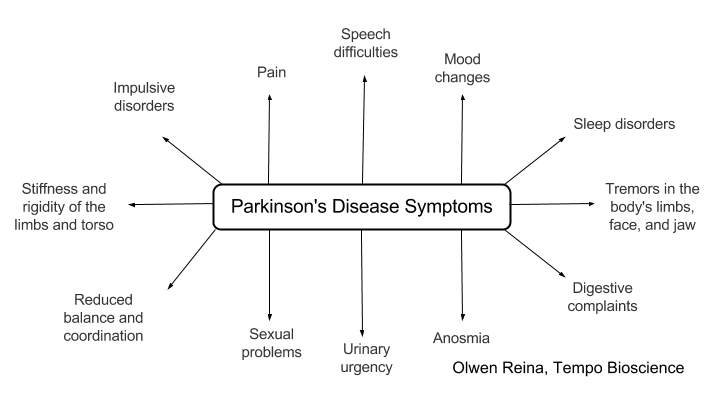
Some of these sounding familiar?
The primary part of the brain affected by this is the substantia nigra, in particular the dopaminergic neurons. As the disease progresses, more and more of the dopamine-producing neurons of the midbrain die so dopamine levels plummet and the above symptoms worsen.
The hallmark of PD are clumps of a protein inside of dopaminergic neurons. These clumps, or Lewy bodies, are composed of a protein called alpha-synuclein. The degradation of these cells explains most of the symptoms you see above. But what about the those not covered?
Interestingly, dopaminergic neurons are not only found in the midbrain, they’re also found in the brainstem and olfactory bulb. Not only that, the intestines also have dopaminergic neurons that degenerate in PD. These two facts may explain the nonmotor and gastrointestinal symptoms of PD.
Since dopamine deficiencies are causing so many problems, the obvious targets for treating PD are to administer dopamine, dopamine precursors and dopamine agonists (trick the brain into thinking it’s getting dopamine) as well as drugs to prevent the breakdown of dopamine precursors (levodopa) in body, called MAO-B inhibitors and COMT inhibitors. Other options treat some of the symptoms of PD like anticholinergics for the tremors.
As with many other disorders, current research seeks to explore the use of stem cells as a therapy in a process known as stem cell engineering. Dopaminergic neurons derived in vitro offer a tantalizing solution to PD: if the problem is a lack of functional neurons, grow some more and give them to the patient. This is known as cell replacement therapy or regenerative medicine. Thus far, however, preclinical tests in rodent models have shown widely varying results. It is thought that the cells produced may be contaminated with other cells or may not be differentiated with high enough fidelity when compared to normal dopaminergic neurons.
It is hoped that recent research showing previously unknown small molecules that regulate the step-wise differentiation of human pluripotent stem cells into dopaminergic neurons as well as new markers to distinguish between dopaminergic neurons and other cells will enable future researchers to achieve more consistent results and finally give us a viable treatment option that stops the progression of or even reverses PD. In addition, it gives us a population of cells that’s a far better facsimile to the real thing for use in preclinical drug testing to screen for new therapies.
